Pyrolysis is a thermochemical decomposition of organic material at elevated temperatures in the absence of oxygen. Pyrolysis typically occurs under pressure and at operating temperatures about 430 °C (800 °F). Pyrolysis is a special case of thermolysis, and is most commonly used for organic materials, being, therefore, one of the processes involved in charring. In general, pyrolysis of organic substances produces gas and liquid products and leaves a solid residue richer in carbon content. Extreme pyrolysis, which leaves mostly carbon as the residue, is called carbonization. The process is used heavily in the chemical industry, for example, to produce charcoal, activated carbon, methanol, and other chemicals from wood, to convert ethylene dichloride into vinyl chloride to make PVC, to produce coke from coal, to convert biomass into syngas, to turn waste into safely disposable substances, and for transforming medium-weight hydrocarbons from oil into lighter ones like gasoline.
Pyrolysis is a thermochemical decomposition of organic material at elevated temperatures in the absence of oxygen. Pyrolysis typically occurs under pressure and at operating temperatures about 430 °C (800 °F). Pyrolysis is a special case of thermolysis, and is most commonly used for organic materials, being, therefore, one of the processes involved in charring. In general, pyrolysis of organic substances produces gas and liquid products and leaves a solid residue richer in carbon content. Extreme pyrolysis, which leaves mostly carbon as the residue, is called carbonization. The process is used heavily in the chemical industry, for example, to produce charcoal, activated carbon, methanol, and other chemicals from wood, to convert ethylene dichloride into vinyl chloride to make PVC, to produce coke from coal, to convert biomass into syngas, to turn waste into safely disposable substances, and for transforming medium-weight hydrocarbons from oil into lighter ones like gasoline.
These specialized uses of pyrolysis may be called various names, such as dry distillation, destructive distillation, or cracking.
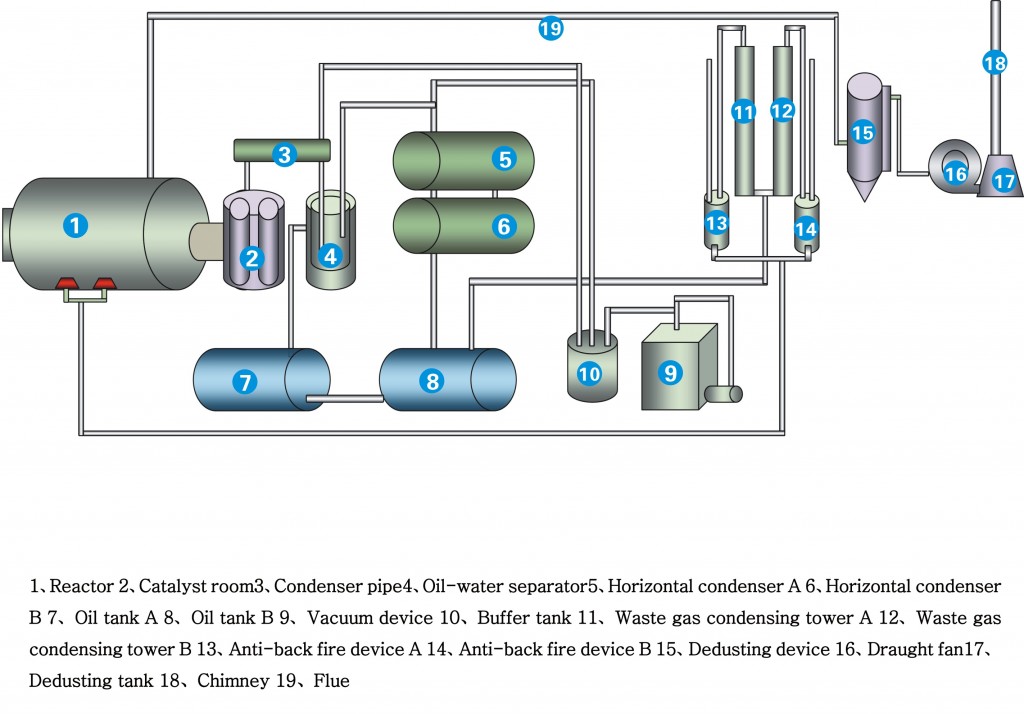
The following is our 5th generation Pyrolysis Plant used for recycling of plastic and tires.